Multitargeted Mechanisms of Curcuma longa in Chronic Disease: Bridging Ethnopharmacology and Evidence-Based Metabolic and Inflammatory Therapy
Published in IJPHDT, Vol-3, Issue-04, April-2026 (Vol. 3, Issue 4, 2026)
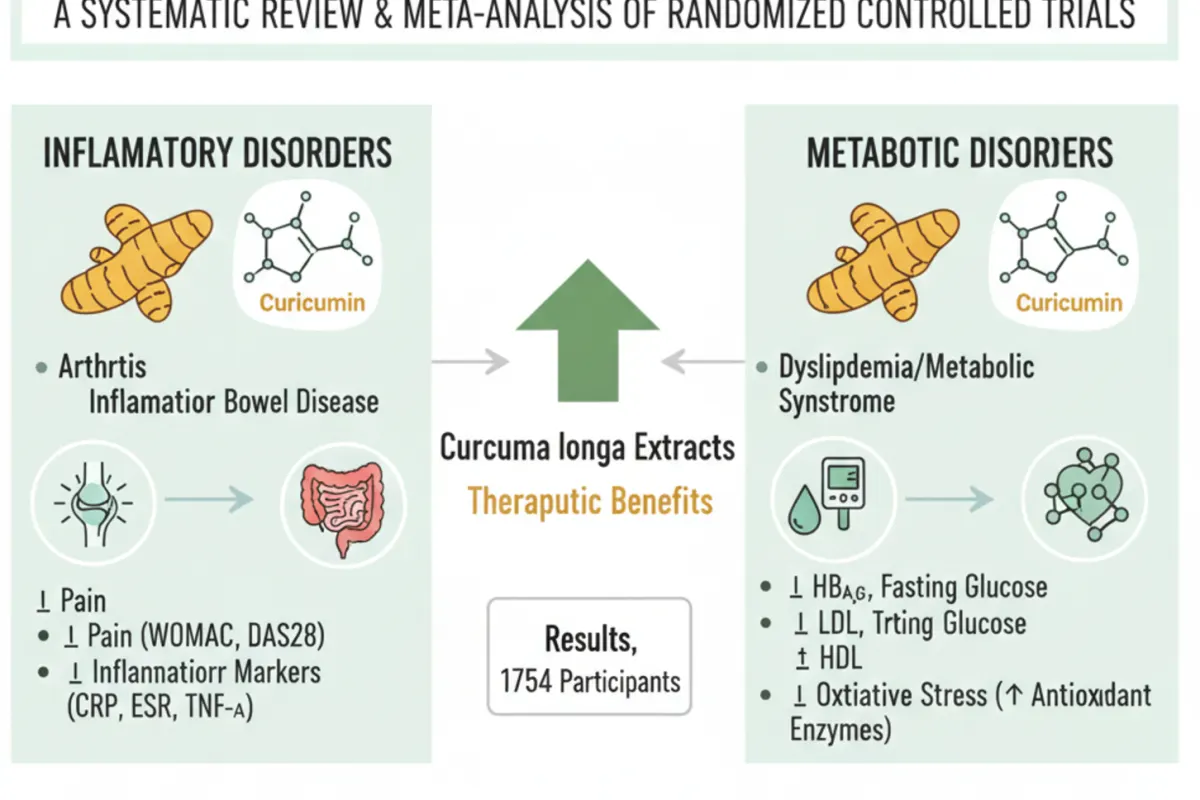
Keywords
Abstract
References
- [1]1. Chandran B, Goel A. A randomized, pilot study to assess the efficacy and safety of curcumin in patients with active rheumatoid arthritis. Phytother Res. 2012;26(11):1719–25. doi:10.1002/ptr.4639
- [2]2. Amalraj A, Gopi S, Varma K, Prasad S, Kunnumakkara AB, et al. Curcumin-galactomannan complex (CurQfen®) benefits in rheumatoid arthritis: a randomized double blind placebo controlled trial. Phytother Res. 2018;32(11):2300–10. doi:10.1002/ptr.6100
- [3]3. Belcaro G, Cesarone MR, Dugall M, Pellegrini L, Ledda A, et al. Meriva® (curcumin phospholipid) in osteoarthritis: randomized double blind study. J Nat Prod. 2014;77(3):545–53. doi:10.1021/np400749v
- [4]4. Henrotin Y, Clutterbuck AL, Allaway D, Lodwig EM, Harris P, et al. A randomized trial of bio optimized curcumin for knee osteoarthritis (Arthrocur®). J Pain Res. 2019;12:975–88. doi:10.2147/JPR.S195467
- [5]5. Panahi Y, Majeed M, Sahebkar A. Curcumin + piperine in metabolic syndrome: a randomized trial. Phytother Res. 2017;31(7):1141–9. doi:10.1002/ptr.5820
- [6]6. Rahimnia AR, Panahi Y, Karimi MH, Dadvar V, Sahebkar A. Curcumin adjunct to standard therapy in inflammatory bowel disease: short term randomized controlled trial. Phytother Res. 2016;30(5):1025–31. doi:10.1002/ptr.5598
- [7]7. Javadi M, Khadem Haghighian H, Goodarzy S, Abbasi M, Nassiri Asl M. Nanomicelle curcumin in rheumatoid arthritis: randomized controlled trial. Int J Rheum Dis. 2019;22(10):1857–62. doi:10.1111/1756-185X.13688
- [8]8. Hodaei H, Adibian M, Nikpayam O, et al. Curcumin supplementation improves insulin resistance and oxidative stress in type 2 diabetes mellitus: randomized controlled trial. Diabetol Metab Syndr. 2019;11:41. doi:10.1186/s13098-019-0437-7
- [9]9. Mansour A, Rahimi HR, Gerami H, et al. Nanocurcumin for diabetic neuropathy: randomized controlled trial. Nutr J. 2025;24:115. doi:10.1186/s12937-025-01184-8
- [10]10. Bateni Z, Rahimi HR, Hedayati M, Afsharian S, et al. Nano curcumin effects on metabolic syndrome: double blind randomized controlled trial. Phytother Res. 2021;35(7):3945–53. doi:10.1002/ptr.7109
- [11]11. Madhu K, Chanda K, Saji MJ. Safety and efficacy of Curcuma longa extract in knee osteoarthritis: randomized controlled trial. Inflammopharmacology. 2013;21(2):129–36. doi:10.1007/s10787-012-0163-3
- [12]12. Ziaei R, Askari G, Moradi S, et al. Curcumin and glycemic control: meta analysis of randomized controlled trials. Adv Exp Med Biol. 2021;1291:139–49. doi:10.1007/978-3-030-56153-6_8
- [13]13. Pourhabibi Zarandi F, Rafraf M, Zayeni H, Asghari Jafarabadi M, et al. Curcumin supplementation in women with rheumatoid arthritis: randomized controlled trial. Phytother Res. 2022;36(4):1797–806. doi:10.1002/ptr.7422
- [14]14. Kulkarni MP, Shakeel A, Shinde BS, Rosenbloom RA. Curcumin extract for knee osteoarthritis: placebo controlled randomized controlled trial. Am J Ther. 2011;18(2):170–7. doi:10.1097/MJT.0b013e318209df49
- [15]15. Hashemzadeh K, Davoudian N, Jaafari MR, Mirfeizi Z. Nanocurcumin in knee osteoarthritis: randomized controlled trial. Curr Rheumatol Rev. 2020;16(2):158–64. doi:10.2174/1874471013666191223152658
- [16]16. Ezzat SM, Khairy ME, Mady FM, et al. Curcumin plus methotrexate in rheumatoid arthritis: a controlled trial. Phytother Res. 2019;33(5):1396–1404. doi:10.1002/ptr.6308
- [17]17. Lau ET L, et al. Nano curcumin adjunct to metformin in type 2 diabetes mellitus: randomized controlled trial. BMC Complement Med Ther. 2020;20:15. doi:10.1186/s12906-019-2801-4
- [18]18. Neta GC, et al. Piperine enhanced Curcuma longa in Brazilian patients with type 2 diabetes mellitus: randomized controlled trial. Nutr Metab Cardiovasc Dis. 2021;31(6):1809–19. doi:10.1016/j.numecd.2021.02.004
- [19]19. Chuengsamarn S, Rattanamongkolgul S, Luechapudiporn R, Phisalaphong C, Jirawatnotai S. Curcumin extract for prevention of type 2 diabetes: a randomized, double blind, placebo controlled trial in prediabetes. Diabetes Care. 2012;35(11):2121–7. doi:10.2337/dc12 0116
- [20]20. Poolsup N, Suksomboon N, Kurnianta PDM, Deawjaroen K. Effects of curcumin on glycemic control and lipid profile in prediabetes and type 2 diabetes mellitus: a systematic review and meta analysis. PLoS One. 2019;14(4):e0215840. doi:10.1371/journal.pone.0215840
- [21]21. Chuengsamarn S, et al. Curcumin extract improves beta cell function and insulin sensitivity in obese T2DM patients: randomized controlled trial. Diabetologia. 2024;67(5):950–9. doi:10.1007/s00125 023 06190 y
- [22]22. Zhang B, Liu K, Yang H, et al. Gut microbiota: the potential key target of TCM’s therapeutic effect of treating different diseases using the same method ulcerative colitis and type 2 diabetes mellitus as examples. Front Cell Infect Microbiol. 2022;12:855075. doi:10.3389/fcimb.2022.855075
- [23]23. Soleimani V, Sahebkar A, Hossein H. Turmeric (Curcuma longa) and its major constituent (curcumin) as nontoxic and safe substances: a review. Phytother Res. 2018;32:985–95. doi:10.1002/ptr.6054
- [24]24. Chainani Wu N, Silverman S, Reingold A, et al. A randomized, placebo controlled, double blind clinical trial of curcuminoids in oral lichen planus. Phytomedicine. 2007;14(5):437–46. doi:10.1016/j.phymed.2007.05.006
- [25]25. Pivari F, Mingione A, Brasacchio C, Soldati L. Curcumin and type 2 diabetes mellitus: prevention and treatment. Nutrients. 2019;11(8):1837. doi:10.3390/nu11081837
- [26]26. Jiménez Osorio AS, Castro Quintero D, Tapia E, et al. Curcumin supplementation in diabetic and non diabetic chronic kidney disease: effects on lipoperoxidation and renal function. Clin Nutr ESPEN. 2020;36:127–33. doi:10.1016/j.clnesp.2019.12.014
- [27]27. Jurgenka JW, et al. Nanocurcumin and rheumatoid arthritis: immunologic outcomes from double blind randomized controlled trial. Int J Rheum Dis. 2019;22(10):1857–62. doi:10.1111/1756 185X.13688
- [28]28. Gu Y, Niu Q, Zhang Q, Zhao Y. Ameliorative effects of curcumin on type 2 diabetes mellitus. Molecules. 2024;29(12):2934. doi:10.3390/molecules29122934
- [29]29. Alsulaim AK, Almutaz TH, Albati AA, Rahmani AH. Therapeutic potential of curcumin in prevention of streptozocin induced diabetes by modulation of oxidative stress and inflammation. Molecules. 2023;29(1):128. doi:10.3390/molecules29010128
- [30]30. Pathomwichaiwat T, et al. Effects of turmeric supplementation on glucose metabolism in diabetes and metabolic syndrome: an umbrella review. PLoS One. 2023;18(7):e0288997. doi:10.1371/journal.pone.0288997
Authors (5)
Prabha Sahu
Shri Rawatpura Sarkar Institut...Shri Rawatpura Sarkar Institute of Pharmacy, Kumha...Shri Rawatpura Sarkar Institute of Pharmacy, Kumhari & Near Power Grid...Shri Rawatpura Sarkar Institute of Pharmacy, Kumhari & Near Power Grid Corp, Behind Holiday Resort, ...
View all publications →Laxmi Sen
Shri Balaji Institute of Pharm...Shri Balaji Institute of Pharmacy and Research Cen...Shri Balaji Institute of Pharmacy and Research Centre, Raipur, 492014Shri Balaji Institute of Pharmacy and Research Centre, Raipur, 492014
View all publications →Aditya Surana
College & address - Royal Coll...College & address - Royal College of Pharmacy, Rai...College & address - Royal College of Pharmacy, Raipur, Chhattisgarh, I...College & address - Royal College of Pharmacy, Raipur, Chhattisgarh, India
View all publications →Renuka Soni
Faculty of Pharmaceutical Scie...Faculty of Pharmaceutical Sciences, Junwani, Bhila...Faculty of Pharmaceutical Sciences, Junwani, Bhilai, Chhattisgarh, Ind...Faculty of Pharmaceutical Sciences, Junwani, Bhilai, Chhattisgarh, India
View all publications →Harsh Kumar Tamrakar
Kamla Institute of Pharmaceuti...Kamla Institute of Pharmaceutical Sciences, Junwan...Kamla Institute of Pharmaceutical Sciences, Junwani, Shri Shankarachar...Kamla Institute of Pharmaceutical Sciences, Junwani, Shri Shankaracharya Professional University, Bh...
View all publications →Download Article
Best for printing and citation
Download Article
Best for printing and citation
Article Information
IJPHDT430009
IJPHDT-01-000240
81-95
2026-04-24
Article Impact
Smart Citations
scite shows how a scientific paper has been cited by providing the context of the citation, a classification describing whether it supports, mentions, or contrasts the cited claim, and a label indicating in which section the citation was made.
How to Cite
Sahu & Sen & Surana & Soni & Kumar, H. (2026). Multitargeted Mechanisms of Curcuma longa in Chronic Disease: Bridging Ethnopharmacology and Evidence-Based Metabolic and Inflammatory Therapy. International Journal of Pharmacognosy and Herbal Drug Technology, 3(4), 81-95. DOI:https://doi.org/10.64063/3049-1630.vol3.issue4.000240
Article Actions
More from this Issue
Beyond the Monthly Cycle: Examining Typical Menstrual Complications, Herbs Management and Their Effects on Female Health
Multitargeted Polyherbal Formulation for Diabetes-Associated Depression: Physicochemical Characterization and In-Vitro Safety Assessment
Role of Herbals in Decoding Complexities of Schizophrenic Brain, Neuroimaging and Genetic Biomarkers
More by These Authors
Standardization and Quality Control of Polyherbal Formulations Using HPLC and UV-Vis Spectroscopy
2025 • Vol. 2, Issue 8
Read more →A Comprehensive Analysis of Herbal Medicine's Possible Use in The Management of Chronic Kidney Disease
2025 • Vol. 2, Issue 1
Read more →Assessment of In-vivo Anti-inflammatory properties of extracts of Sida cordata
2025 • Vol. 2, Issue 10
Read more →
